Clinical trial recruitment today identifies who qualifies biologically — but fails to account for whether a person is able, willing, or prepared to participate. Gryt bridges that gap.
CTPA augments diagnosis-based eligibility with lived-experience intelligence — to activate, and sustain real clinical trial participation.
Over the past decade, Gryt's innovative CTPA solution has successfully enrolled high-priority trials and helped clients achieve FDA goals.
Eligibility Criteria Miss the Full Picture
Static inclusion and exclusion criteria identify who qualifies biologically — but ignore whether a patient is able, willing, or prepared to participate. This diagnosis-only approach drives costly enrollment delays, protocol amendments, and early dropouts.
Traditional Approach
Clinical Eligibility
Diagnosis, biomarkers, and clinical attributes determine who qualifies biologically for a trial.
What's Missing
Real-World Participation
Lived experience, behavioral readiness, and life circumstances determine who actually enrolls and stays.
Clinical Trial Patient Activation (CTPA)
Gryt's proven model augments traditional eligibility criteria with lived-experience and behavioral readiness factors — moving from identification to activation.
Diagnosis-Only Model
Who qualifies biologically?
- Focuses on diagnosis, biomarkers, and clinical attributes
- Static inclusion and exclusion criteria only
- Does not account for lived experience or readiness
- High rates of non-enrollment, early disengagement, and dropout
- Costly recruitment delays and protocol amendments
Gryt's CTPA Model
Who will enroll, engage, and complete?
- Augments clinical criteria with lived-experience intelligence
- Identifies behavioral readiness and participation barriers
- Surfaces actionable interventions to increase activation
- Improves retention across the full trial lifecycle
- Especially effective for precision medicine, rare disease, and hard-to-reach populations
Outcome
By integrating lived-experience intelligence with clinical eligibility, CTPA enables participation-ready recruitment — reducing enrollment delays, improving retention, and increasing trial success.
10+
Years Activating Patients
2×
SCOPE Award Winner
FDA
Goals Achieved for Clients
A Decade of Proven Results
After working with Lilly COI and leading LIVESTRONG's Clinical Trial Matching Service, David Craig co-founded Gryt Health in 2016 to close the gap between eligibility and real participation.
Gryt Health Founded
Co-founded by David Craig following experience at Lilly COI and leading LIVESTRONG's Clinical Trial Matching Service.
First Client: Ignyta — NTRK Trial
Gryt's first client engagement successfully supported enrollment of Ignyta's landmark NTRK clinical trial.
SCOPE Participant Engagement Award
Recognized at SCOPE 2019 for industry-leading work in patient engagement and clinical trial participation.
SCOPE Award — Second Recognition
Awarded again at SCOPE 2021, cementing Gryt's reputation as a leader in participant-centered trial engagement.
Expanding CTPA Across Precision Medicine
Gryt continues to advance lived-experience intelligence as a core driver of clinical trial success across rare disease, oncology, and underrepresented populations.
Ready to Activate Your Trial?
Let's talk about how Gryt's CTPA model can reduce enrollment delays, improve retention, and help your trial succeed — especially for hard-to-reach populations.
Clinical Trial Patient Activation (CTPA)
GRYT’s “CTPA” is a proven solution to drive recruitment and support representation on high-priority clinical trials.
We developed our innovative approach through successful collaboration with sponsors to:
Position clinical trials with clearly articulated differentiators
Increase awarenesss with physicians and patients based on targeted geo and demographic factors – particularly for underrepresented patients
Utilize a scalable approach with real-time engagement and conversion analytics
Demonstrate results and optimize efforts with bi-weekly reporting
Over the past decade, Gryt’s innovative CTPA solution has successfully enrolled high-priority trials and helped our clients achieve FDA goals.
After working with Lilly COI and leading LIVESTRONG’S Clinical Trial Matching Service, David Craig co-founded Gryt Health in 2016.
Gryt’s first client was Ignyta, where we helped successfully enroll its NTRK trial
Gryt was awarded the Participant Engagement Award at SCOPE in 2019 and 2021
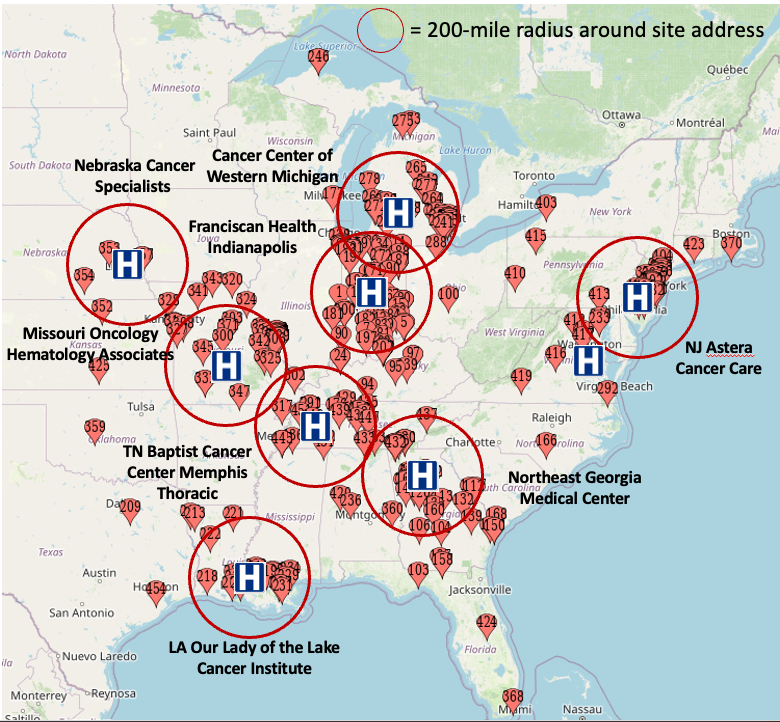
Engagement by Site Location
Each pin represents a person searching on Google, saw the campaign, utilized Gryt’s engagement platform and then took action for enrollment.
Outcomes Achieved
Facilitated patient-focused trial education, boosting awareness and screening
Engaged underrepresented patients by race, ethnicity & rural settings
Delivered actionable insights/ analytics for overall and site-specific recruitment
Increased patient diversity in screening
KPI’s are utilized to complete the read-out for FDA Diversity Action Plan